
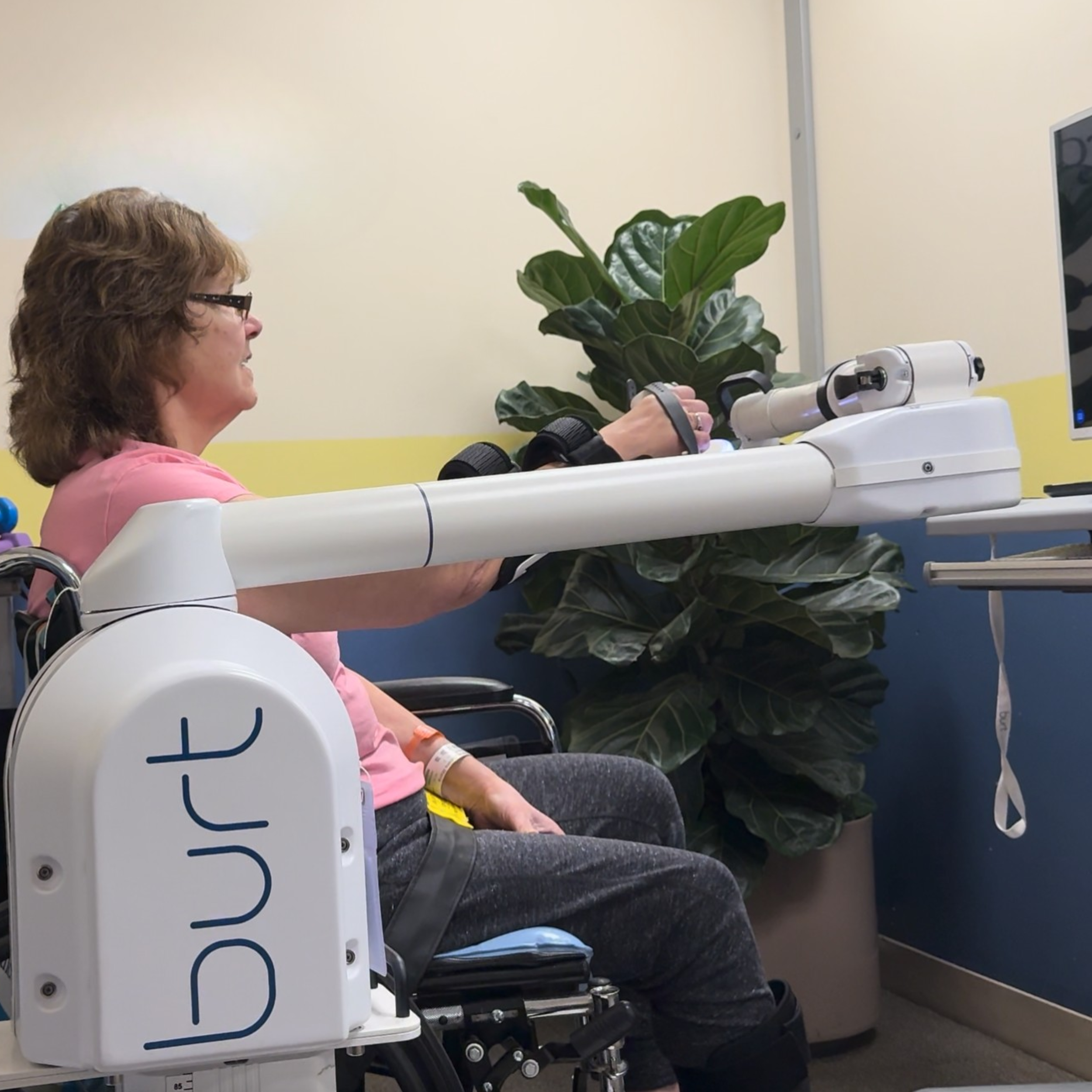
Burt® is an upper-limb therapy robot.
Patients using Burt® regain mobility through realistic, haptic activities. It allows a patient to float weightlessly while applying gentle forces to resist, assist, and give subtle queues.
Burt® is a sense of hope.
New Beginnings
John is a fisherman.
But after an unexpected stroke just before Christmas in 2024, John spent 30 days in a San Antonio hospital.
During his stay, John utilized Burt® daily to regain mobility in his right arm. Through Barrett's FishFood and PirateCatcher activities, he built strength, progressing from BurtAssist® to BurtResist® in just 4 weeks.
John walked out excited to get back on the water.
“Burt® makes you more enthusiastic about getting well ... I’d recommend it to anybody!”

Burt® is Barrett's FDA-registered, Class II robotic rehabilitation device
Burt® is for upper-limb therapy following stroke, TBI, and other neurological conditions.
Used in rehab facilities across the country, Burt® combines backdrivable haptic technology with realistic activities of daily living.
Burt® is Better
60-second Setup
---
18 Realistic Activities
---
Haptic Feedback
---
3 Planes of Backdrivability
---
Cognitive Challenges
---
Strength Assessments
---
60-second Setup --- 18 Realistic Activities --- Haptic Feedback --- 3 Planes of Backdrivability --- Cognitive Challenges --- Strength Assessments ---
We’re powering the future of motion. Here’s how:
60-second setup - with quick left/right swap
18 activities - including our IADL game, LaundryLoader
Haptic feedback during therapy
Offloads weight - Burt® supports the patient’s arm
Patient safety - backdrivable in the transverse, sagittal, and frontal planes
Cognitive challenges - attention, memory, visual neglect activities
Strength assessments - set goals and track progression
And many more… get the FREE brochure to learn more
-
Burt® is designed for patients recovering from stroke, traumatic brain injury (TBI), and other neurological conditions affecting upper-limb mobility.
The device supports rehabilitation across the transverse, sagittal, and frontal planes, helping patients rebuild strength, coordination, and range of motion through realistic activities.
-
Yes. Burt® is an FDA-registered, ISO-certified Class II medical device engineered and assembled at Barrett's facility in Newton, Massachusetts. It is sold worldwide through Barrett's network of partners.
-
Most rehab robots require extensive setup time and offer limited patient interaction. Burt® sets up in about 60 seconds with a quick left/right arm swap, and patients engage with realistic haptic activities like sorting laundry or catching objects rather than abstract exercises.
Burt® is fully backdrivable, meaning it can never force a patient's arm into an unsafe position, which also eliminates physical strain on the therapist.
-
A therapist selects an activity and difficulty level, adjusts Burt® to assist or resist based on the patient's current ability, and the patient interacts with the game on the display by moving the device's arm.
Sessions can target strength, coordination, cognitive function, or a combination. After each session, Burt® generates a progress score so therapists can track improvement over time.
-
Yes. Burt® includes built-in strength assessments and scoring after every session. Therapists can set goals, monitor progression across sessions, and use the data to adjust treatment plans or report outcomes to administrators and insurers.




